- Numeric improvements were seen across key multiple sclerosis measures, including the Modified Fatigue Impact Scale, Expanded Disability Status Scale, Timed 25-Foot Walk test, Pyramidal Scores and NeuroQoL™ Fatigue scores.
- Data was presented at ECTRIMS (39th Congress of the European Committee for Treatment and Research of Multiple Sclerosis) on October 11, 2023
NEW YORK, October 16, 2023 -- Tiziana Life Sciences Ltd. (Nasdaq: TLSA) (“Tiziana” or the “Company”), a biotechnology company developing breakthrough immunomodulation therapies via novel routes of drug delivery, today announced six-month data showing positive clinical improvements related to Modified Fatigue Impact Scale (MFIS) scores and similar important clinical measures of physical function in foralumab-treated, non-active Secondary Progressive Multiple Sclerosis (na-SPMS) patients participating in an Expanded Access (EA) Program. This follows on from previously announced positive six-month PET scan data which was presented at ECTRIMS 2023.
The findings, which are summarized in table 1 below, show broad based six month improvements across various key measures for multiple sclerosis. Secondary progressive multiple sclerosis is hallmarked by an increase of disability over time. The table below shows a stabilization or an improvement in physical function of the various clinical measures over a six-month period.
Table 1
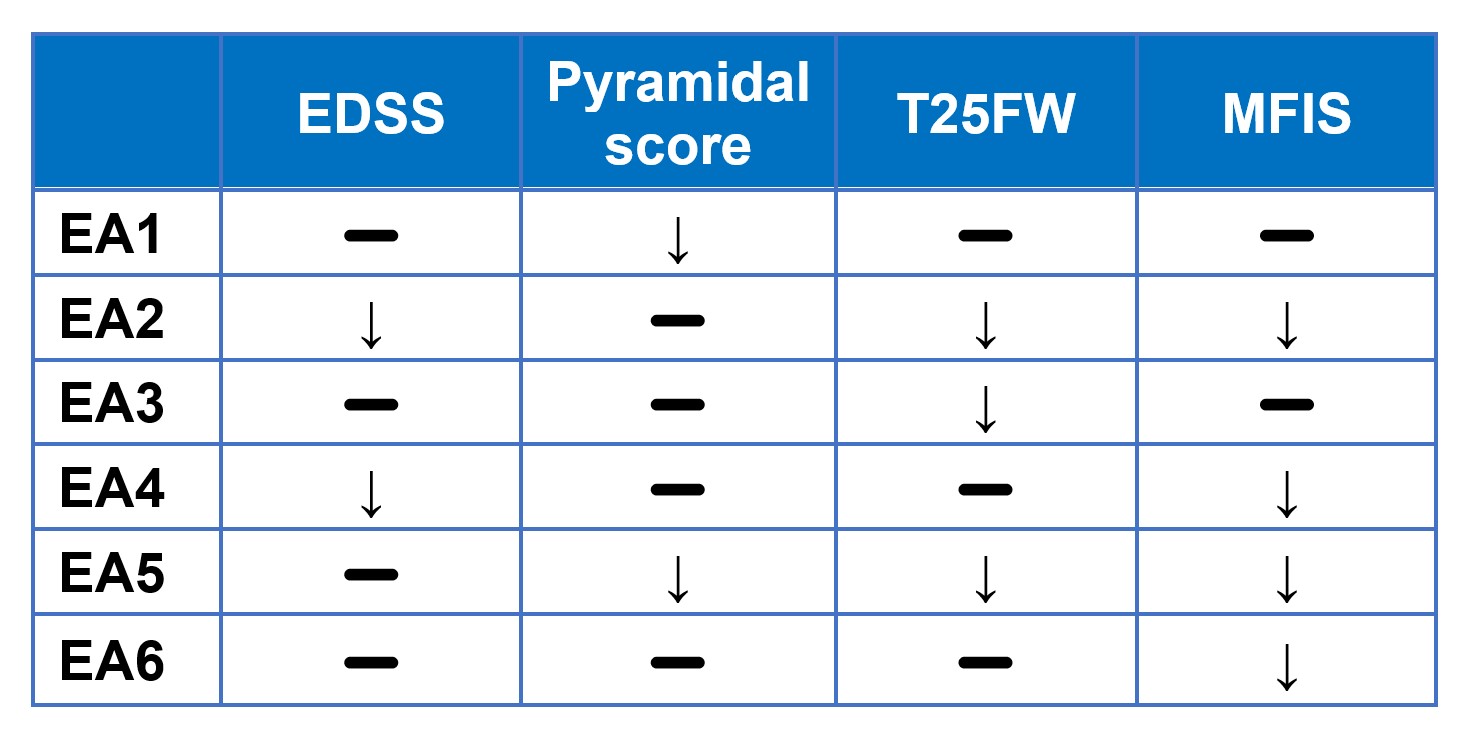
- Denotes stabilization
↓ Denotes improvement
Fatigue, as measured above in MFIS, refers to an overwhelming sense of physical, mental, and emotional exhaustion that is disproportionate to the level of activity or effort exerted. It is a major, common, and often debilitating symptom experienced by many individuals with MS. It differs from the typical tiredness that everyone experiences from time to time. In the context of MS, it is called ‘primary fatigue’ and is a direct result of damage to the central nervous system. This kind of fatigue can significantly impact a person’s daily life and functioning.
“I am pleased to see the continued clinical response to intranasal foralumab from patients enrolled in our expanded access na-SPMS program,” stated Dr. Tanuja Chitnis, M.D., Principal Investigator and Professor of Neurology at Harvard Medical School (HMS) and senior neurologist at Brigham and Women’s Hospital, a founding member of Mass General Brigham Healthcare System. “Notably, in this latest clinical update for patients EA 3 through EA 6, we have seen improvement in the Modified Fatigue Impact Scale scores in three out of four patients, which is significant since fatigue is a major complaint in this population. We have also seen various degrees of improvement in the Expanded Disability Status Scale, Timed 25-Foot Walk Test, pyramidal function scores and NeuroQoL Fatigue scores in a disease state that typically shows a decline in function over time.
Howard L. Weiner, M.D., Chairman of Tiziana’s Scientific Advisory Board and Co-Director of the Ann Romney Center for Neurologic Diseases at Brigham and Women’s Hospital noted, “With six patients now dosed in our na-SPMS Expanded Access Program and followed over a six-month period, we are seeing a highly encouraging efficacy signal in this difficult-to-treat patient population.”
Gabriele Cerrone, Chairman, acting CEO and founder of Tiziana Life Sciences noted “Intranasal foralumab is continuing to show clinical improvements over time in patients with na-SPMS where foralumab targets inflammation in the brain. Based on the totality of the clinical results seen to-date, I am hopeful that intranasal foralumab and its anti-inflammatory mechanism of action could provide relief to na-SPMS patients that currently have no available therapies.”
About Foralumab
Activated T cells play an important role in the inflammatory process. Foralumab, the only fully human anti-CD3 monoclonal antibody (mAb), binds to the T cell receptor and dampens inflammation by modulating T cell function, thereby suppressing effector features in multiple immune cell subsets. This effect has been demonstrated in patients with COVID and with multiple sclerosis, as well as in healthy normal subjects. The non-active SPMS intranasal foralumab Phase 2 trial is expected to start screening in November of 2023. Immunomodulation by nasal anti-CD3 mAb represents a novel avenue for treatment of neuroinflammatory and neurodegenerative human diseases.[1],[2]
About Tiziana Life Sciences
Tiziana Life Sciences is a clinical-stage biopharmaceutical company developing breakthrough therapies using transformational drug delivery technologies to enable alternative routes of immunotherapy. Tiziana’s innovative nasal approach has the potential to provide an improvement in efficacy as well as safety and tolerability compared to intravenous (IV) delivery. Tiziana’s lead candidate, intranasal foralumab, which is the only fully human anti-CD3 mAb, has demonstrated a favorable safety profile and clinical response in patients in studies to date. Tiziana’s technology for alternative routes of immunotherapy has been patented with several applications pending and is expected to allow for broad pipeline applications.
For further inquiries:
Tiziana Life Sciences Ltd
Paul Spencer, Business Development and Investor Relations
+44 (0) 207 495 2379
email: info@tizianalifesciences.com
Investors:
Irina Koffler
LifeSci Advisors, LLC
+1 646 970 4681
ikoffler@lifesciadvisors.com
[1] https://www.pnas.org/doi/10.1073/pnas.2220272120
[2] https://www.pnas.org/doi/10.1073/pnas.2309221120