Anti Interlukin-6 Receptor mAB
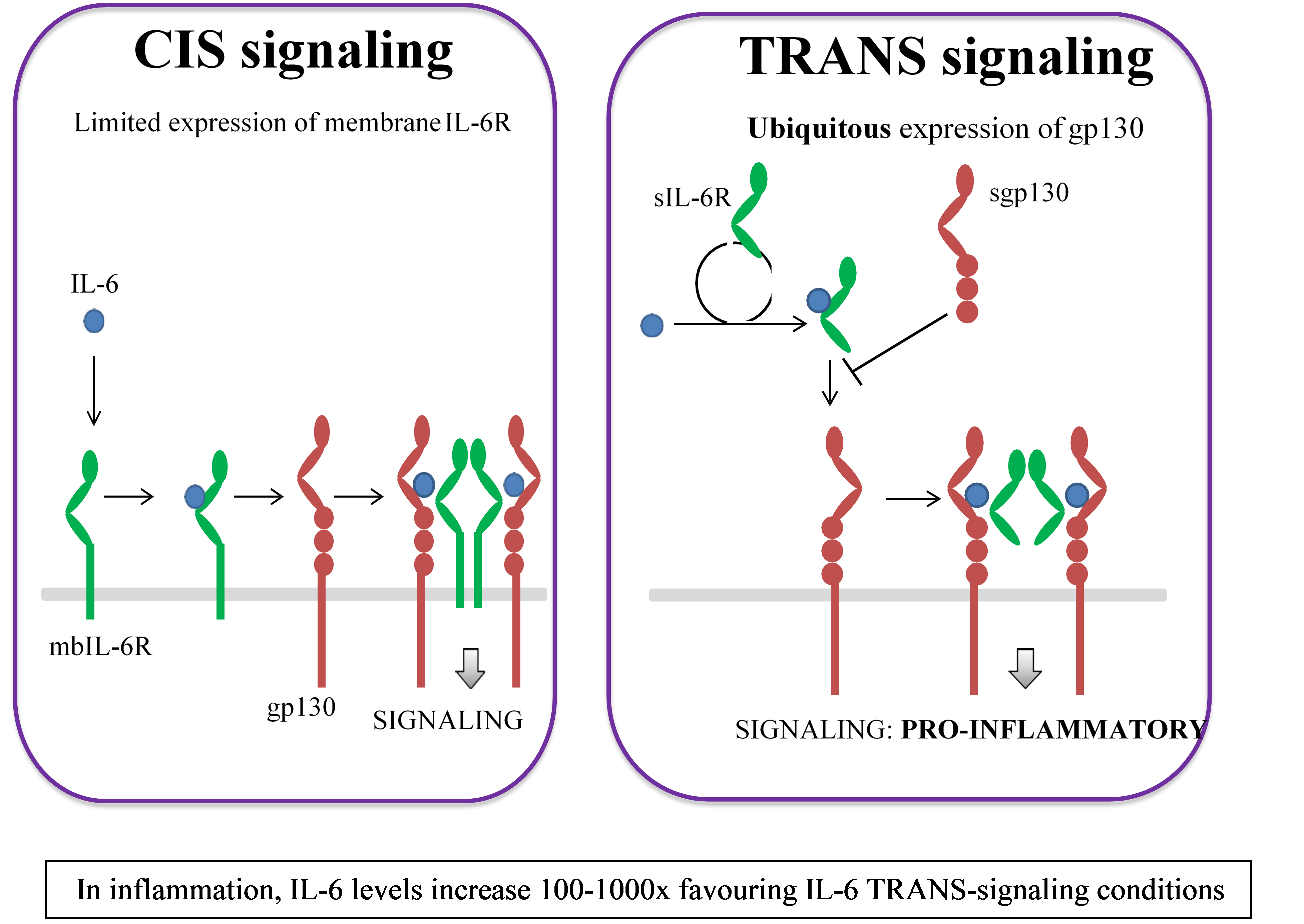
Tiziana's Anti IL-6R mAb (TZLS-501), a fully human mAb binds to both membrane-bound and soluble forms of IL-6R, an inflammatory cytokine driving chronic inflammation associated with autoimmune disease and cancer, reducing circulating levels of the IL-6 cytokine. Anti-IL-6R antibody can potentially be used in combination with Foralumab or other anti-inflammatory and anti-infective agents as therapy for idiopathic pulmonary fibrosis (IPF), acute respiratory distress syndrome (ARDS), multiple myeloma, arthritis, lupus and oncology indications. Excessive production of IL-6 is regarded as a key driver of chronic inflammation and is believed to be associated with severe lung damage and chronic fibrosis observed with acute and chronic respiratory illness.
The Company is scaling GMP manufacturing of its anti-IL-6R mAb concurrently with developing a hand-held nebulizer technology for direct delivery of the antibody into the for treatment of patients with IPF, a rare disease indication. Manufacturing of clinical supplies for a Phase 1 study is expected soon.